Emery Haley, PhD, Scientific Writing Specialist
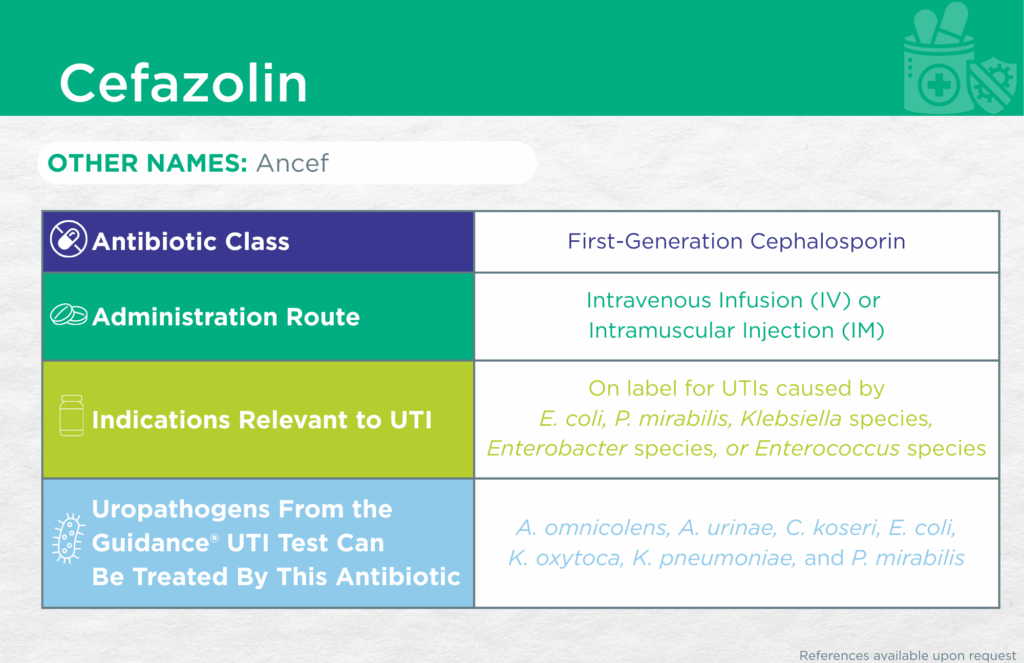
Cefazolin
Find the Latest FDA-Approved Labelling Information Here: Drugs@FDA Online Database
Administrative Routes
Parenteral [intravenous infusion (IV) or intramuscular injection (IM)]
Other Names
Ancef
Bacteriostatic or Bactericidal
Bactericidal [1]
Antibiotic Class
First-Generation Cephalosporin
Mechanisms of Action
Cefazolin, a beta-lactam antibiotic, binds to penicillin-binding proteins (PBPs) on bacterial cell walls. PBPs are essential for the formation of peptidoglycan, which gives the bacterial wall strength and integrity. Binding of cefazolin to PBPs leads to failure of peptidoglycan cell wall synthesis, causing bacterial cell death.
WHO AWaRe Classification
Access [2]
Empiric Use Recommendations
No published guidance
Indication(s) Relevant to UTI
On label for UTIs caused by Escherichia coli, Proteus mirabilis, Klebsiella species, Enterobacter species, and Enterococcus species.
Checkmarks
CLSI and/or FDA documents support the efficacy of this antibiotic against the following organisms from the Guidance® UTI test: Aerococcus urinae, Citrobacter koseri, Escherichia coli, Klebsiella oxytoca, Klebsiella pneumoniae, and Proteus mirabilis
Published primary literature supports the efficacy of this antibiotic against the following organism from the Guidance® UTI test: Alloscardovia omnicolens [3]


